A Nurse Is Teaching a Client Who Has a Family History of Colorectal Cancer
Abstract
Background
Antibody-induced gut dysbiosis has been associated with colorectal cancer (CRC) in older adults. This study will investigate whether an association exists between antibiotic usage and early-onset colorectal cancer (CRC), and as well evaluate this in afterward-onset CRC for comparison.
Methods
A instance-control study was conducted using master intendance data from 1999–2011. Assay were conducted separately in early-onset CRC cases (diagnosed < 50 years) and later on-onset cases (diagnosed ≥ 50 years). Provisional logistic regression was used to calculate odds ratios and 95% confidence intervals (CI) for the associations between antibiotic exposure and CRC past neoplasm location, adjusting for comorbidities.
Results
Seven thousands nine hundred and three CRC cases (445 aged <50 years) and 30,418 controls were identified. Antibiotic consumption was associated with colon cancer in both historic period-groups, particularly in the early-onset CRC accomplice (<50 years: adapted Odds Ratio (ORadj) 1.49 (95% CI 1.07, 2.07), p = 0·018; ≥50 years (ORadj (95% CI) 1.09 (i.01, 1.18), p = 0·029). Antibiotics were not associated with rectal cancer (<50 years: ORadj (95% CI) 1.17 (0.75, 1.84), p = 0.493; ≥50 years: ORadj (95% CI) ane.07 (0.96, 1.19), p = 0.238).
Conclusion
Our findings propose antibiotics may take a role in colon tumour formation beyond all age-groups.
Background
Since the tardily 1980s, global antibiotic consumption and cases of early-onset colorectal cancer (CRC) have increased markedly [1,ii,3]. This design may be related; antibiotic consumption has been associated with CRC genesis in adults of all ages [four,5,6,vii,eight]. In contrast to declining incidence amid older populations [5, nine], CRC incidence among adults aged 20–29 years in Europe is increasing by ~8% each twelvemonth [1]. In the Us, CRC is the second well-nigh mutual incident cancer and tertiary leading crusade of cancer decease in adult males less than fifty years old [ten]. Consensus exists that early-onset CRC (<fifty years) is different to later-onset CRC (≥50 years) in terms of epidemiology, pathology and biology [5, 6, xi], although more contempo show suggests both types are clinically and genomically indishtinguishable [12]. Therefore, at that place may be a rationale for studying early-onset CRC separately from afterward-onset CRC to identify specific risk factors associated with the rising trend observed among younger people.
Worldwide, in that location were ~70 billion doses of antibiotics consumed in 2011—which equates to 10 per person on earth [thirteen]. Although essential for many medical interventions, children and teenagers are amongst those most commonly exposed to antibiotic therapy [14] and may be more vulnerable to the potential furnishings of overexposure—such equally obesity, allergic diseases and inflammatory bowel disease [v, 15]. In the USA, 69% of children anile less than 2 years are exposed to antibiotics [16], and every bit accessibility to antibiotics increases beyond depression and middle-income countries, antibody usage for common childhood infections is becoming more than widespread [17, 18]. Furthermore, loftier prevalence of acne amongst adolescents tin can event in long-term antibiotic exposure, sometimes lasting months to years due to varying national guidelines and uncertainty regarding optimum treatment elapsing [19]. In addition, at least xx–xxx% of antibiotics prescribed in primary care may be inappropriate [18, 20].
The human relationship between pathogenic organisms and cancer is well-established; Helicobacter pylori is associated with gastric cancer and human papilloma virus (HPV) with anal, cervical, tonsillar and vulval cancer [21, 22]. Antibiotic-induced microbiome changes can be permanent and irregularities in immunostimulatory bacterial products tin impede normal immune-surveillance, increasing the chance of carcinogenesis [21]. In addition, interruption of normal gut commensals may allow colonisation past pathogenic bacteria, which invade and damage the gut mucosa, leading to inflammation and neoplasm germination [22]. Examples of these harmful microbes include strains of Escherichia. coli and Bacteroides. fragilis; which may exist promoted by certain antibiotics [22, 23].
Several single-centred studies [four, 24,25,26,27,28,29,xxx,31,32] and two recent systematic reviews and meta-analyses [7, 8] found an association betwixt oral antibiotics CRC take chances. However, these studies are contradictory; whether risk varies with tumour location or with use of anti-aerobic or anti-anaerobic agents (i.e. antibiotics active confronting bacteria requiring oxygen, or lack of oxygen, to survive) or both, remains uncertain. To our cognition, no study before has investigated antibiotic usage in the early on-onset CRC population; participant mean age is ~69–72 years in all recorded studies [4, 24,25,26,27,28,29,30,31,32].
This written report seeks to determine the association betwixt antibiotic utilize and early on-onset CRC, and whether any run a risk may differ within the colorectal continuum, or by antibody spectrum of activity.
Materials and methods
Data source
Study data were obtained from the population-based Chief Care Clinical Information Unit Research (PCCIUR) database [33], comprising over two 1000000 patients registered at 393 general practices beyond Scotland betwixt 1993 and 2011. PCCIUR contains upwardly to 20 years of demographic, clinical and diagnostic information and has been widely used in epidemiological research [34,35,36,37].
Study design
A case-command study was conducted using PCCIUR data. Cases were patients with a new diagnosis of primary CRC (Read codes B13, B14, run into Supplementary Material Table S1) between 1999 and 2011. Cases were excluded if they had a previous cancer, excluding non-melanoma skin cancer, or were diagnosed with other primary cancers on the date of diagnosis due to doubt about the principal cancer and the potential for coding errors. Cases of anal cancer were excluded as they are squamous prison cell cancers and associated with HPV infection. Patients with diagnosed conditions predisposing to CRC (e.g. inflammatory bowel illness, Peutz-Jeghers syndrome, polyposis syndromes) were excluded equally our report was limited to sporadic CRC. Patients with diagnosed immunosuppressive states (due east.g. Sjogren'due south syndrome, HIV infection, transplantation) and those in receipt of immunosuppressive medicines during the exposure flow (come across definition below) were also excluded.
All available controls (alive, registered with a GP and free from cancer (excepting non-melanoma peel cancer)) were identified for each case matching on practice, year of birth (±5 years), gender and yr of registration (in categories). Up to v controls for each case were randomly selected from those bachelor, without replacement. The alphabetize date within each matched set was defined as the diagnosis date of CRC in the example. Both cases and controls needed at least 3 years of follow-up data and remained registered with the same general exercise over the follow-up menstruation. Two strata were constructed for comparative purposes, a younger strata (cases plus matched controls <fifty years) and an older strata (cases plus matched controls ≥ 50 years) [5, 6, 38]. Information extraction are depicted in Fig. 1.
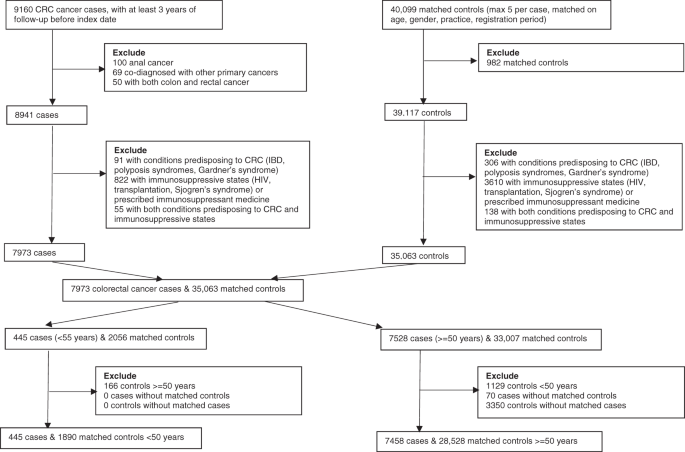
CRC: colorectal cancer; IBD: inflammatory bowel disease.
Inside each matched set, the exposure period, i.e. the period of fourth dimension over which medicine apply was adamant, started on either 1 January 1993 (equally prescriptions before this time were unlikely to be recorded electronically), or the most contempo GP registration date if this occurred after Jan 1993. This ensured all members within each matched set had the same exposure period. The exposure period concluded 1 year earlier the index date, to reduce the take chances of reverse causality and exclude medications unlikely to accept had sufficient fourth dimension to cause cancer [39]. Rectal or rectosigmoid junction tumours were classified equally rectal cancer, otherwise tumours were classified as colon cancer.
Classification and definition of antibiotic exposure
Prescriptions for oral antibiotics were extracted from PCCIUR. These were classified past drug course and by presence or absence of anti-anaerobic effects to provide insight into bacterial populations potentially associated with CRC [4]. Medicines studied are listed in the Supplementary Textile Table S2.
For each antibiotic prescription, the elapsing of treatment (in days) was identified from prescribing records. Where this was not recorded (n = 536 (1.i%) of all antibiotic prescriptions), handling duration was estimated according to standard dosing for each antibiotic. Total exposure in days of all antibiotic classes was calculated for each patient and categorised as 0 days, ane–15 days, 16–60 days, >60 days [40, 41]. Analyses were also conducted using cumulative duration of anti-anaerobic or non-anti-anaerobic antibiotic treatment. For these analyses only master clinical therapeutic effect(s) of each medicine were considered; other antimicrobial activity is often less pronounced without major effects on aerobic or anaerobic populations [42].
Covariates
The following comorbidities, based upon published Read codes for the Charlson Comorbidity Index (CCI) [43], were identified prior to or during the exposure period: diabetes, myocardial infarction, coronary heart illness, middle failure, peripheral vascular disease, dementia, cerebrovascular disease, chronic obstructive pulmonary disease, osteoporosis, renal disease, liver disease and hemiplegia/paraplegia. Additional comorbidities, relevant to CRC (i.eastward. gallstones, acromegaly), were likewise identified. We also adjusted for use of low dose aspirin and not-steroidal anti-inflammatory drugs (NSAIDs), equally these may reduce hazard of CRC [44, 45]. Smoking status (not-smoker, current smoker, onetime smoker) [46] and alcohol consumption (non-drinker, low-cal or moderate drinker, heavy drinker) [47] were determined from the virtually recent smoking or alcohol tape prior to or during the exposure period.
Statistical analysis
Descriptive statistics summarised cases and controls. For each cohort, conditional logistic regression was used to calculate odds ratios (OR) and 95% confidence intervals (CI) for associations between each exposure and CRC, with adjustment for comorbidities. The matched design accounted for age (±5 years), GP practice, gender and yr of registration. All analyses were adjusted for age in years, every bit participants were matched in age bands rather than by calendar twelvemonth. Interaction tests to adamant whether antibiotic exposure effects varied by strata. To test for tendency in risk of colorectal cancer beyond different categories of treatment length, the duration of antibody exposure was treated as a continuous rather than a categorical variable. Associations betwixt individual classes of antibiotics and colon/rectal cancer are reported as supplementary analyses due to low prescribing levels of private classes among patients under fifty years and the increased risk of type 1 errors due to multiple testing.
Subgroup analyses
Analyses were repeated for matched sets where location of cases' colon tumour was explicitly recorded in the diagnostic readcodes, namely proximal colon (cancerous neoplasms of hepatic flexure, transverse colon, caecum, appendix or ascending colon) and distal colon (cancerous neoplasms of the descending colon, sigmoid colon or splenic flexure of colon). The master analyses were repeated using the subsample of patients with recorded body mass index (BMI).
Sensitivity analyses
Sensitivity analyses were undertaken as follows: (1) period of time before index date during which prescriptions were not counted was increased from 1 to 2 years to reduce potential for opposite causation; (two) threshold used to distinguish between younger and older patients was lowered from 50 years to 45 years; (iii) threshold used to distinguish between younger and older patients was increased from l years to 55 years; (4) adjustments were fabricated for comorbidities, smoking and booze use for the 23,702 patients (61.2%) where both lifestyle factors had been recorded in the patient's clinical records. The latter assay was also repeated using multiple imputation with chained equations (MICE) techniques to impute smoking and booze status. This is a simulation-based method appropriate for handling missing data assuming that such values are missing at random. Ordered logit models were used with age, gender, deprivation within the GP practice locality, and comorbidities for the imputations, stratified by case-control status and using 25 imputations [48].
Results
Descriptive statistics: cases and controls
Seven thousands ix hundred and three CRC cancer cases and thirty,418 matched controls were identified. Five thousands iii hundred fifty half-dozen cases (67.8%) had at least four matched controls. There were 5281 colon cancer cases and 2662 rectal cancer cases. Median (inter-quartile range (IQR)) age at diagnosis in the younger and older strata was 45 [41, 47] years and 71 years (63, 78), respectively. The exposure menstruum, matched in cases and controls, was slightly shorter for patients <50 years (median (IQR) 6.9 (four.8, 9.2) years) than patients ≥ fifty years (median (IQR) vii.ix (v.3, x.8) years). Approximately 55% of patients were male in each age-grouping.
Characteristics of cases and controls are listed in Tabular array one. A full set of descriptive statistics of cases and controls by each neoplasm location is provided as Supplementary Fabric Tabular array S3.
Descriptive statistics: antibiotic medication
44.9% (17,206) of patients were prescribed antibiotics during the exposure period. The proportion of CRC cases prescribed antibiotics was larger than the proportion of controls prescribed antibiotics in both the <50 strata (cases: 47.2% (210) v controls: 40.1% (757)) and the ≥ 50 years strata (cases: 46.eight% (3496) v controls: 44.vii% (12,743)). Near commonly prescribed antibiotics were penicillins (52.8% (25,473) of all antibiotic prescriptions). The proportion of cases prescribed each class of antibody was usually higher than the proportion of controls in both historic period-groups for both colon and rectal cancer.
Antibiotics with anti-anaerobic effects were more commonly prescribed than antibiotics without anti-anaerobic issue (52.7% (25,440) 5 47.3% (22,851)). Prescribing of both anti-anaerobic antibiotics and non-anti-anaerobic antibiotics was college amid cancer cases than controls in both historic period-groups and cancer sites. Descriptive statistics for class of antibiotic medication by age-group and tumour location are given in Table 2.
Associations between use of antibiotics and CRC
Use of antibiotics was associated with increased risk of colon cancer in both age-groups (<l years: adjusted Odds Ratio (ORadj) 1.49 (95% CI 1.07, 2.07), p = 0·018; ≥fifty years: ORadj 1.09 (95% CI one.01, one.18), p = 0.029)) (Table three). Although this effect was greater among patients <fifty years compared to those ≥ fifty years, the difference was non significant (interaction examination: p = 0.071). At that place was no apparent exposure-response relationship between antibiotic use and colon cancer chance for either age-groups (<fifty years: P-trend p = 0.177; ≥50 years: P-trend p = 0.082). Antibiotics apply was not significantly associated with increased rectal cancer risk in either age-group (<50 years: ORadj one.17 (95% CI 0.75, 1.84), p = 0.493; ≥50 years: ORadj 1.07 (95% CI 0.96, 1.19), p = 0·238)).
Analyses of antibiotic prescribing by treatment elapsing and CRC location are depicted graphically in Forest Plots (Fig. 2) and listed in Supplementary Textile Table S4.
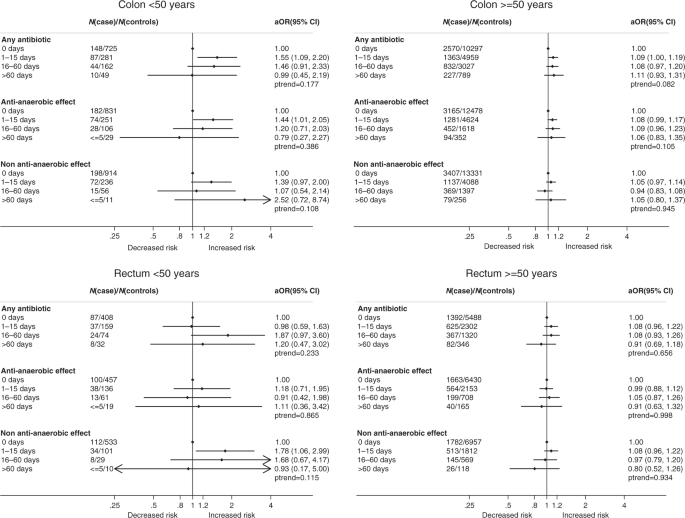
aOR: adjusted Odds Ratio; N: Number analyses adjusted for diabetes, myocardial infarction, coronary center disease, heart failure, peripheral vascular disease, dementia, cerebrovascular affliction, chronic obstructive pulmonary illness, osteoporosis, renal disease, liver disease, hemiplegia/paraplegia, gallstones, acromegaly, low dose aspirin and NSAIDs
There was no evidence at the 5% statistical significance level of any differences between the the 2 age-groups in associations between classes of antibiotic utilize and the take a chance of colon or rectal cancer (Supplementary Textile Table S5).
Associations between anti-anaerobic furnishings and CRC
Although not significant, employ of anti-anaerobic antibiotics was associated with increased risk of colon cancer in both strata (<fifty years: ORadj i.34 (95% CI 0.97, 1.86), p = 0.078; ≥50 years: ORadj 1.08 (95% CI 1.00, one.17), p = 0.054) (Table 3). However, these effects did not differ significantly from each other (interaction test: p = 0·204) and there was no credible exposure-response relationship in either age-group (<50 years: P-tendency p = 0.386; ≥50 years: P-trend p = 0.105). Antibiotic use was not significantly associated with increased adventure of rectal cancer in either age-group (<50 years: ORadj i.11 (95% CI 0.seventy, 1.76), p = 0.646; ≥50 years: ORadj one.00 (95% CI 0.89, i.11), p = 0.974)).
Associations between non-anti-anaerobic furnishings and CRC
There was some prove that employ of antibiotics without anti-anaerobic effects was associated with increased gamble amongst patients <50 years of both colon cancer (ORadj 1.37 (95% CI 0.97, 1.93), p = 0·075) and rectal cancer (ORadj ane.·70 (95% CI 1.06, 2.74), p = 0.029), merely not among patients ≥ fifty years (colon cancer: ORadj 1.03 (95% CI 0.95, i.11), p = 0.500; rectal cancer: ORadj 1.·05 (95% CI 0.93, 1.18), p = 0·436) (Table iii). However, these effects neither vary betwixt age-groups (interaction test: colon cancer p = 0.151, rectal cancer p = 0.888), nor was in that location an exposure-response relationship between use of antibiotics without anti-anaerobic effects and either colon or rectal cancer risk in either age-groups.
Subgroup analyses
There were 687 (13.0%) colon cancer cases classified as proximal colon and 551 (10.4%) classified equally distal colon. Use of antibiotics, antibiotics with anti-anaerobic furnishings and antibiotics without anti-anaerobic effects was associated with increased risk of proximal colon cancer among <50 s (any antibiotic ORadj 3.78 (95% CI 1.sixty, 8.92), p = 0.002; anti-anaerobic ORadj two.17 (95% CI 1.00, iv.68), p = 0.049; non-anti-anaerobic ORadj 2.86 (95% CI 1.21, 6.73), p = 0·016). It appeared effects associated with antibiotic utilise and anti-anaerobic antibiotics differed between the two historic period-groups (interaction examination: whatever antibiotic p = 0.001; anti-anaerobic p = 0.034). A positive exposure-response relationship was too observed between antibiotic prescribing and take chances of proximal colon cancer among the younger patients (P-trend = 0.004)). Results for both subgroup analyses are listed in Table three and Supplementary Material Table S6.
One-3rd of all patients included in our analyses (n = 12,657) (33·0%) had their BMI reported, and these patients were on boilerplate slightly overweight (median (IQR) 26.7 (23.9,29.9)). Patients with recorded BMI were less likely to be non-smokers or non-drinkers, and accept higher reported levels of comorbidities and prescribed medication, than patients where BMI was missing (Supplementary Material Table S7). Adjusting for BMI in improver to comorbidities and medicine use increased the magnitude of the association between whatsoever antibiotic use and early-onset colon cancer risk (ORadj i.98 (95% CI 0.82, 4.81), p = 0.130), although this association did not differ significantly betwixt that reported with the older age-grouping (ORadj i.01 (95% CI 0.87, 1.16), p = 0.920) (interaction test: p = 0.139). Full details of these subgroup analyses are reported in Supplementary Material Tables S8 and S9.
Sensitivity analyses
Results from sensitivity analyses are listed in Table 4. Increasing lag-time from 1 year to 2 years or additionally adjusting for alcohol and smoking had no substantive impact on reported associations between antibiotic use and CRC take chances.
Discussion
In this big population-based instance-command study of early on-onset CRC cases and later-onset CRC cases, antibody consumption was associated with colon cancer pathogenesis across all age-groups.
Results from a systematic review and meta-analysis of 10 high-quality observational studies plant antibiotic utilize increased CRC take chances (issue size (ES) 1.17 (95% CI 1.05, ane.30)), just associations differed with tumour location and antibiotic classes [seven]. Analysis of colon cancer cases alone showed no significant association (ES 1.06 (95% CI 0.89, i.26)). However, in that location was high heterogeneity between studies (I2 = 95.seven% and 83.v%, respectively), which—if our findings are true—may partly reverberate varying and older age-groups included in those studies. Other than colon and rectal cancer, the meta-analysis did not explore the influence of antibiotics on neoplasm locations further—such as association with proximal colon cancer. Another systematic review and meta-assay suggested a weak clan may exist between antibiotic consumption and run a risk of CRC [8]. Nevertheless, definitive conclusions cannot be made given the small number of studies included, a lack of control for misreckoning and high heterogeneity. Furthermore, none of the studies analysed antibiotic exposure during babyhood and adolescence, a time when individuals are most vulnerable to gut dysbiosis [eight].
Although we found limited associations between antibiotic usage and rectal cancer across all age-groups, not-anti-anaerobic (i.due east. exclusively anti-aerobic) antibiotics among the young age-group but were observed to increment adventure of colon, rectal, proximal and distal colon cancer more than anti-anaerobic antibiotics. This conflicts with a example-command written report of participants anile twoscore–70 years, which constitute anti-aerobic antibiotics to protect against distal colon and rectal cancer, whereas anti-anaerobic antibiotics increased chance of cancer—especially in the proximal colon [4]. A further study [28] found both anti-aerobic and anti-anaerobic agents were associated with CRC, whereas another found just anti-anaerobic antibiotics increased risk [27]. However, sample sizes amongst our early-onset CRC cohort were small, especially when stratified into not-anti-anaerobic antibiotics and length of treatment. Furthermore, information technology may be clinically irrelevant whether anti-anaerobic or anti-aerobic antibiotics have a role in tumour formation, as about antibiotic drugs have dual anti-anaerobic and anti-aerobic action.
Congruent with existing studies, a very strong association was observed betwixt antibiotic consumption and proximal colon cancer [four, 40, 49]; however, this was only observed in the early-onset CRC subgroup assay, which had a small sample size of just l cases. With a greater microbial diversity and concentration of short-concatenation fatty acids, the proximal colon is more than vulnerable to antibiotic exposure than the distal colon and rectum [49, 50]. Dysbiosis results in contradistinct bacterial activity, fermentation and therefore colonic pH, in addition to suspension of protective colonic mucus leading to straight contact between the biofilm and epithelial cells, leading to chronic inflammation [12, 50, 51]. In all cohorts, at that place was limited evidence of a positive exposure-response human relationship between cumulative antibiotic use and risk of CRC, with the exception of proximal colon cancer in the younger accomplice. This supports previous literature suggesting take a chance increases later on minimal antibiotic use [four], with risk not necessarily increasing with prolonged antibiotic exposure [7, 49].
Whether the observed relationship between antibiotics and CRC is causal remains uncertain. CRC is a circuitous, "heterogenous" disease with many underlying molecular mechanisms and risk factors [xi]. Compared to later-onset illness, early on-onset CRC has been described every bit a remarkably distinctive subset of illness [5, 6, 11]. Therefore, to compare our findings with previous studies, which take not considered the impact of age on CRC in improver to antibiotic exposure, may be inappropriate. If we were to condone this fact, co-ordinate to the Bradford Hill criteria [52], it is probable that a causal relationship may exist. Our findings point a strong association betwixt antibiotic use and CRC, especially with colon cancer. Our study is somewhat consistent with the literature, suggesting a human relationship does exist—even if outcome sizes vary. In that location is evidence of temporality in other studies [29], although we found no evidence of a biological gradient except in the case of early-onset proximal colon cancer. A causal human relationship is plausible and coherent, and we can describe parallels with other normally accustomed phenomena—such as antibiotic-induced microbiome changes increasing adventure of obesity, autoimmune disease and metabolic disorders [53,54,55], and the anticancer effects of a healthy microbiome [21]. Nonetheless, the relationship is not particularly specific; with around 67 million courses of antibiotics prescribed each year in the USA to children aged less than 19 [18], exposure to antibiotics among the young is incredibly common. Information technology is therefore difficult to judge how many of these exposed individuals will potentially be diagnosed with early-onset CRC—a relatively rare illness outcome [9].
In our study, we observed more participants with CRC had rectal cancer in the younger rather than the older cohort. A study investigating U.s.a. early-onset CRC trends suggest rectal cancer incidence in the young is increasing more rapidly than colon cancer; by 2030, they predict incidence of colon and rectal cancer will increase by 90% and 124% among patients aged 20–34 years [56]. A possible clan may exist between sexually transmitted infections and early-onset rectal cancer; [49] Chlamydia infections have malignant potential and secondary rectal infection is mutual [57, 58]. A review of clinical and molecular features of early-onset CRC suggests distal colon and rectal cancer are predominantly features of early-onset CRC, whereas proximal cancers tend to characteristic in later-onset disease [xi]. Despite this, it is likely that sporadic early and later-onset CRC are otherwise indistinguishable in terms of genomics and biology [12]. The embryological origins of the proximal colon (midgut) and distal colon and rectum (hindgut) are different, as are biological features of cancers arising in these areas; proximal have more microsatellite instability, and distal more than chromosomal instability [59]. Together with differences in the luminal contents and microbiome, information technology is biologically plausible that antibiotic consumption could influence the development of colonic and rectal cancer differentially past location.
There are multiple elements likely to be driving the increase in early-onset CRC including dietary factors—such as increased consumption of scarlet and candy meat, monosodium glutamate, titanium dioxide and loftier-fructose corn syrup; obesity; stress; reduced exercise; and antibiotic consumption [five]. There is a scarcity of studies investigating early-life exposures and adult-onset cancers, although the aforementioned factors at interplay are known to have adverse effects on the microbiome. In improver, lifestyle changes occurring since the 1950s correlate with the increased rates of CRC, specially among the young [lx]. Evidence of possible carcinogenic effects of antibiotics are express [61], even so some antibiotics commonly up-regulate cyclooxygenase-2—a mechanism proven to promote development of CRC [62, 63]. Furthermore, information technology is the antibiotic-induced microbiome changes which disrupt immunostimulatory bacteria and requite ascension to pathogenic colonisation which is probable to exist carcinogenic, rather than the actual medications themselves [21, 22].
This study has several strengths. PCCIUR is nationally representative, roofing at least xv% of the Scottish general practise population [35]. Comprehensive linking of practise data to Scottish Cancer Registry data provides high coverage of CRC cases (given the relative rarity of early-onset CRC) and a relatively long exposure menses. Thorough cleaning and validation of the information has minimised loss of prescription items due to transcription errors. This allowed accurate calculation of cumulative antibody exposure in primary care by grade or spectrum of action. In the Britain, antibiotics can just be obtained with a medical prescription, and over-the-counter purchases are not possible. Although nosotros could not access secondary care prescriptions, antibiotics commenced in hospital with long-term intent will announced in subsequent GP prescribing records. In our analyses we brand a distinction between patients with early on-onset CRC and subsequently-onset CRC, and used sensitivity analysis to determine whether results inverse when the historic period threshold used to define the ii strata was altered.
Inevitably, this study also has its limitations. Given CRC in those anile less than 50 years is relatively rare, we had merely 445 cases. Our sample size decreased farther when nosotros explored antibody spectrum of activeness and specific neoplasm locations, and inevitably some analyses amongst patients under 50 years will be underpowered. Individuals with immunosuppressing weather condition or diagnosed genetic predispositions to CRC were excluded; these make up a significant proportion of early-onset CRC patients, and the touch of antibiotic therapy in these groups will therefore not exist measurable. Although we managed to exclude participants with genetic predispositions to CRC, the PCCIUR dataset does not provide information on a participants' family history or dietary habits (BMI was only reported for ~33% of our sample). These both have a significant influence on CRC gamble, for example the increased gamble associated with obesity and CRC is well-known [64]. However the low numbers of cases among patients under 50 years where BMI was reported means that we cannot annotate substantively on the nature of whatever association of BMI with CRC take a chance on the footing of our analyses. Long-term effects of exposure to antibiotics in childhood, when the gut microbiome is developing and potentially more vulnerable, are all the same to exist evaluated in terms of cancer risk and may exist of clinical importance [65]. Unfortunately, lack of prescribing data in PCCIUR prior to 1993 ways we are not able to explore whether initial age of exposure to antibiotics is associated with CRC risk.
Our dataset is suspectable to various biases associated with observational data, such equally differential think and contrary causation. The latter may be observed if patients presenting with stomach hurting are initially diagnosed with gastrointestinal infection and prescribed antibiotics. Although we adapted for use of medicines, smoking and alcohol utilize, residual misreckoning may exist present (e.1000. NSAID strength/duration of prescribing, units of alcohol consumed, number of pack years for smokers). There may be unmeasured confounding in the main analyses due to the inability to adjust for other relevant confounders not reported comprehensively in our data (eastward.thou. BMI). Discrepencies in tumour location data may exist, with cases recorded as 'colon' rather than the sub-site within the colon. In addition, some patient groups will be missing from primary care records and cannot be accounted for, such equally the homeless, individual patients and prisoners. There volition too be variability between prescribers regarding completeness of comorbidity recording. Data not captured past PCCIUR includes most secondary care prescriptions, individual healthcare records, antibiotic prescriptions earlier 1993 (as these volition not have been recorded electronically), and prescription adherence. These could exist highly relevant to the study. Finally we cannot guarantee that patients adhered to their prescription medication, which seems likely to dilute whatever real associations, although studies suggest adherence to antibiotic therapy is high [66].
Our findings, showing no significant deviation between the associations with early and later-onset illness, supporting recent bear witness suggesting there are more similarities than differences between early on and afterwards-onset illness [12]. Therefore future studies to further elucidate any role of antibiotics in CRC genesis should exist inclusive of all age-groups.
In conclusion, our findings advise antibiotic exposure is associated with CRC genesis across all age-groups. Information technology is possible that antibiotic exposure may be contributing to cases of CRC, potentially more then amidst the young. Our study raises the question whether antibiotic usage history should exist included in the standardised proformas for referral from master to secondary intendance. Further studies to confirm our findings and evaluate long-term effects of antibiotics on gut health are required, and increased awareness of the potential harms associated with antibiotic usage among clinicians and members of the public is necessary.
Information availability
The datasets analysed in this study are not publicly bachelor and were used under license. Requests for PCCIUR data should be directed in the first example to Katie Wilde (Research Manager), electronic mail: grand.wilde@abdn.ac.united kingdom of great britain and northern ireland.
Code availability
Standard statistical techniques were used to analyse the data, for which code tin can be obtained via the corresponding writer. All statistical analyses were undertaken using Stata xv [67]. Results were pregnant if they referred to statistical significance on a ii-sided blueprint-based test evaluated at the 5% level.
References
-
Vuik FER, Nieuwenburg SAV, Bardou M, Lansdorp-vogelaar I, Dinis-ribeiro Thou, Bento MJ, et al. Increasing incidence of colorectal cancer in young adults in Europe over the concluding 25 years. Gut. 2019;68:1820–vi.
-
Irish potato CC, Singal AG, Baron JA, Sandler RS, Simmons HC, Cancer C, et al. Decrease in Incidence of Young-Onset Colorectal Cancer Before Recent Increment. Gastroenterology. 2019;155:1716–9.
-
Boeckel TP Van, Gandra Southward, Mpp AA, Caudron Q, Grenfell PBT, Levin PSA, et al. Global antibiotic consumption 2000 to 2010: an analysis of national pharmaceutical sales data. Lancet Infect Dis [Internet]. 2014;14:742–l.
-
Zhang J, Haines C, Watson AJM, Hart AR, Platt MJ, Pardoll DM, et al. Oral antibiotic use and take a chance of colorectal cancer in the Britain, 1989–2012: a matched case-control report. 2019;68:1–8.
-
Hofseth LJ, Hebert JR, Chanda A, Chen H, Love BL, Pena MM, et al. Early-onset colorectal cancer: initial clues and current views. Nat Rev Gastroenterol Hepatol [Net]. 2020. https://doi.org/10.1038/s41575-019-0253-4.
-
Patel SG, Ahnen DJ. Colorectal Cancer in the Young. Curr Gastroenterol Rep. 2018;xx:15. https://doi.org/10.1007/s11894-018-0618-9.
-
Simin J, Fornes R, Liu Q, Olsen RS, Callens South, Engstrand Fifty, et al. Antibiotic use and risk of colorectal cancer: a systematic review and dose–response meta-analysis. Br J Cancer [Internet]. 2020;123:1825–32.
-
Sanyaolu LN, Oakley NJ, Nurmatov U, Dolwani S, Ahmed H. Antibiotic exposure and the risk of colorectal adenoma and carcinoma: a systematic review and meta-analysis of observational studies. Color Dis. 2019;22:858–lxx.
-
Perrott S, Laurie G, Laws Chiliad, Johnes A, Miedzybrodzka Z, Samuel L. Young-onset colorectal cancer in the N East of Scotland: survival, clinico-pathological features and genetics. BMC Cancer [Internet]. 2020;twenty:108.
-
Siegel RL, Miller KD, Goding Sauer A, Fedewa SA, Butterly LF, Anderson JC, et al. Colorectal cancer statistics, 2020. CA Cancer J Clin. 2020;lxx:145–64.
-
Ballester V, Rashtak Southward, Boardman L. Clinical and molecular features of young-onset colorectal cancer. Earth J Gastroenterol. 2016;22:1736–44.
-
Cercek A, Chatila WK, Yaeger R, Walch H, Fernandes GDS, Krishnan A, et al. A Comprehensive comparison of early-onset and average-onset colorectal cancers. J Natl Cancer Inst [Internet]. 2021. https://doi.org/ten.1093/jnci/djab124.
-
Blaser MJ, Melby MK, Lock M, Nichter G. Bookkeeping for variation in and overuse of antibiotics amongst humans. BioEssays. 2021;43:e2000163. https://doi.org/10.1002/bies.202000163. Epub 2021 January 6.
-
Dolk CK, Pouwels KB, Smith DRM, Robotham JV, Smieszek T. Antibiotics in primary intendance in England: which antibiotics are prescribed and for which atmospheric condition? J Antimicrob Chemother. 2018;73:2–10.
-
Butel 1000-J, Waligora-Dupriet A-J, Wydau-Dematteis South. The developing gut microbiota and its consequences for wellness. J Dev Orig Health Dis. 2018;9:590–7. https://doi.org/10.1017/S2040174418000119. Epub 2018 Mar 22.
-
Bailey LC, Forrest CB, Zhang P, Richards TM, Livshits A, DeRusso PA. Clan of antibiotics in infancy with early on babyhood obesity. JAMA Pediatr. 2014;168:1063–9.
-
Allwell-Brown G, Hussain-Alkhateeb L, Kitutu Iron, Strömdahl S, Mårtensson A, Johansson EW. Trends in reported antibiotic use among children nether 5 years of age with fever, diarrhoea, or cough with fast or difficult breathing across low-income and middle-income countries in 2005–17: a systematic analysis of 132 national surveys from 73 countrie. Lancet Glob Heal. 2020;viii:e799–807.
-
Center for Disease Command. Antibiotic utilise in the United States, 2018: Progress and Opportunities. The states Department of Health and Human Services. 2018.
-
Bhate Grand, Lin LY, Barbieri J, Leyrat C, Hopkins South, Stabler R, et al. Is there an association betwixt long-term antibiotics for acne and subsequent infection sequelae and antimicrobial resistance? A systematic review protocol. BMJ Open. 2020;10:e033662.
-
Public Health England. Enquiry reveals levels of inappropriate prescriptions in England [Cyberspace]. 2018. https://www.gov.uk/regime/news/enquiry-reveals-levels-of-inappropriate-prescriptions-in-england.
-
Zitvogel Fifty, Daillère R, Roberti MP, Routy B, Kroemer Chiliad. Anticancer effects of the microbiome and its products. Nat Publ Gr [Internet]. 2017;15:465–78.
-
Whisner CM, Aktipis CA. The part of the microbiome in cancer initiation and progression: how microbes and cancer cells utilize excess energy and promote i another'south growth. Curr Nutr Rep. 2019;eight:42–51.
-
Goodwin Ac, Destefano CE, Wu South, Huso DL, Wu X. Polyamine catabolism contributes to enterotoxigenic Bacteroides fragilis -induced colon tumorigenesis. PNAS. 2011;108:15354–9.
-
Kilkkinen A, Rissanen H, Klaukka T, Pukkala E, Heliövaara M, Huovinen P, et al. Antibody use predicts an increased risk of cancer. Int J Cancer. 2008;123:2152–5.
-
Friedman GD, Coates AO, Potter JD, Slattery 1000. Drugs and colon cancer. Pharmacoepidemiol Drug Saf. 1998;7:99–106.
-
Friedman GD, Jiang SF, Udaltsova N, Quesenberry CP, Chan J, Habel LA. Epidemiologic evaluation of pharmaceuticals with limited testify of carcinogenicity. Int J Cancer. 2009;125:2173–8.
-
Wang J, Chang C. Infection, antibiotic therapy and risk of colorectal cancer: a nationwide nested instance-control study in patients with Type 2 diabetes mellitus. Int J Cancer. 2014;967:956–67.
-
Dik VK, Oijen MGHVan, Smeets HM, Siersema PD. Frequent use of antibiotics is associated with colorectal cancer chance: results of a nested case-control written report. Dig Dis Sci. 2016;61:255–64.
-
Armstrong D, Dregan A, Ashworth Thousand, White P, McGee C, de Lusignan S. The association between colorectal cancer and prior antibiotic prescriptions: case control study. Br J Cancer [Net]. 2020;122:912–7.
-
Didham RC, Reith DM, McConnell DW, Harrison KS. Antibiotic exposure and breast cancer in New Zealand. Breast Cancer Res Treat. 2005;92:163–7.
-
Boursi B, Haynes M, Mamtani R, Yang YX. Impact of antibiotic exposure on the run a risk of colorectal cancer. Pharmacoepidemiol Drug Saf. 2015;24:534–42.
-
Falagas ME, Walker AM, Jick H, Ruthazer R, J G, Snydman DR. Late incidence of cancer after metronidazole user/non-user study. Clin Infect Dis. 1998;26:384–8.
-
University of Aberdeen. Primary Care Clinical Informatics Unit of measurement Research. The Institute of Applied Health Sciences. https://world wide web.abdn.ac.u.k./iahs/research/main-care/pcciur/alphabetize.php. Accessed Appointment 2019-02-18.
-
Busby J, Murchie P, Murray L, Iversen L, Lee AJ, Spence A, et al. The effect of medications which cause inflammation of the gastro-oesophageal tract on cancer risk: a nested case–command study of routine Scottish data. Int J Cancer. 2017;140:1828–35.
-
Tran KT, McMenamin C, Hicks B, Murchie P, Thrift AP, Coleman HG, et al. Proton pump inhibitor and histamine-2 receptor adversary utilise and run a risk of liver cancer in ii population-based studies. Aliment Pharmacol Ther. 2018;48:55–64.
-
Spence AD, Busby J, Murchie P, Kunzmann AT, McMenamin ÚC, Coleman HG, et al. Medications that relax the lower oesophageal sphincter and risk of oesophageal cancer: an analysis of ii independent population-based databases. Int J Cancer. 2018;143:22–31.
-
Macfarlane Television receiver, Lefevre K, Watson MC. Aspirin and non-steroidal anti-inflammatory drug use and the risk of upper aerodigestive tract cancer. Br J Cancer. 2014;111:1825–59.
-
Dozois EJ, Boardman LA, Suwanthanma Westward, Limburg PJ, Cima RR, Bakken JL, et al. Young-onset colorectal cancer in patients with no known genetic predisposition: Can we increase early on recognition and improve outcome? Medicine (Baltim). 2008;87:259–63.
-
Pottegård A, Hallas J. New utilise of prescription drugs prior to a cancer diagnosis. Pharmacoepidemiol Drug Saf. 2017;26:223–7.
-
Cao Y, Wu K, Mehta R, Drew DA, Vocal M, Lochhead P, et al. Long-term use of antibiotics and take a chance of colorectal adenoma. Gut. 2018;67:672–8.
-
Zhang J, Haines C, Watson AJM, Hart AR, Platt MJ, Pardoll DM, et al. Oral antibiotic employ and run a risk of colorectal cancer in the United Kingdom, 1989-2012: a matched case-control study. Gut. 2019;68:1971–8.
-
Rafii F, Sutherland JB, Cerniglia CE. Furnishings of handling with antimicrobial agents on the human colonic microfl ora. Therapeutics and Clinical Risk Management. 2008.
-
Khan NF, Perera R, Harper S, Rose Pw. Adaptation and validation of the Charlson Index for Read/OXMIS coded databases. BMC Fam Pract. 2010;11:1.
-
Garcia-Albeniz Ten, Chan AT. Aspirin for the prevention of colorectal cancer. Best Pract Res Clin Gastroenterol. 2011;25:461–72.
-
Sangha S, Yao M, Wolfe MM. Not-steroidal anti-inflammatory drugs and colorectal cancer prevention. Vol. 81, Postgraduate Medical Journal. The Fellowship of Postgraduate Medicine; 2005. p. 223–seven.
-
Hannan LM, Jacobs EJ, Thun MJ. The Association between cigarette smoking and risk of colorectal cancer in a large prospective cohort from the United States. Cancer Epidemiol Biomarkers Prev. 2009;18:3362–vii.
-
Pedersen A, Johansen C, Grønbæk M. Relations between amount and type of alcohol and colon and rectal cancer in a Danish population based accomplice written report. Gut. 2003;52:861–seven.
-
Raghunathan T, Lepkowski J, Van Hoewyk J, Solenberger P. A multivariate technique for multiply imputing missing values using a sequence of regression models. Surv Methodol. 2001;27:85–95.
-
Lu SSM, Mohammed Z, Häggström C, Myte R, Lindquist E, Gylfe Å, et al. Antibiotics employ and subsequent risk of colorectal cancer: a Swedish Nationwide Population-Based Study. J Natl Cancer Inst. 2021;00:1–ix.
-
Dejea CM, Wick EC, Hechenbleikner EM, White JR, Mark Welch JL, Rossetti BJ, et al. Microbiota organisation is a distinct feature of proximal colorectal cancers. Proc Natl Acad Sci USA [Internet]. 2014;111:18321–six.
-
den Besten G, van Eunen K, Groen AK, Venema Grand, Reijngoud D-J, Bakker BM. The part of brusque-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J Lipid Res [Internet]. 2013;54:2325–40.
-
Fedak KM, Bernal A, Capshaw ZA, Gross S. Applying the Bradford Colina criteria in the 21st century: how data integration has changed causal inference in molecular epidemiology. Emerg Themes Epidemiol. 2015;12:1–9.
-
Kasselman LJ, Vernice NA, DeLeon J, Reiss AB. The gut microbiome and elevated cardiovascular risk in obesity and autoimmunity. Atherosclerosis [Internet]. 2018;271:203–13.
-
Myers B, Brownstone Northward, Reddy 5, Chan S, Thibodeaux Q, Truong A, et al. The gut microbiome in psoriasis and psoriatic arthritis. Best Pr Res Clin Rheumatol [Net]. 2019;33:101494.
-
Sharma Southward, Tripathi P. Gut microbiome and type 2 diabetes: where we are and where to become? J Nutr Biochem [Internet]. 2019;63:101–8.
-
Bailey CE, Hu C-Y, Yous YN, Bednarski BK, Rodriguez-Bigas MA, Skibber JM, et al. Increasing disparities in age-related incidence of colon and rectal cancer in the United States, 1975-2010. JAMA Surg. 2015;150:17–22.
-
Chumduri C, Gurumurthy RK, Zadora PK, Mi Y, Meyer TF. Chlamydia infection promotes host Deoxyribonucleic acid damage and proliferation only impairs the DNA damage response. Cell Host Microbe [Internet]. 2013;xiii:746–58.
-
Heijne JCM, van Liere GAFS, Hoebe CJPA, Bogaards JA, van Benthem BHB, Dukers-Muijrers NHTM. What explains anorectal chlamydia infection in women? Implications of a mathematical model for examination and treatment strategies. Sex Transm Infect [Cyberspace]. 2017;93:270–v.
-
Cisyk AL, Nugent Z, Wightman RH, Singh H, McManus KJ. Characterizing microsatellite instability and chromosome instability in interval colorectal cancers. Neoplasia [Net]. 2018;xx:943–50.
-
Akimoto Northward, Ugai T, Zhong R, Hamada T, Fujiyoshi K, Giannakis M, et al. Rising incidence of early-onset colorectal cancer—a call to activity. Nat Rev Clin Oncol [Net]. 2021;18:230–43.-
-
De MouraM, Van. HoutenB. An update on the genotoxicity and carcinogenicity of marketed pharmaceuticals with reference to in silico predictivity: a review article. Environ Mol Mutagen [Internet]. 2010;405:391–405.
-
Attur MG, Patel RN, Patel PD, Abramson SB, Amin AR. Tetracycline up-regulates COX-2 expression and prostaglandin E2 production independent of its effect on nitric oxide. J Immunol [Internet]. 1999;162:3160–7.
-
Dixon DA, Blanco FF, Bruno A, Patrignani P. Mechanistic aspects of COX-2 expression in colorectal neoplasia. Recent Results Cancer Res. 2013;191:vii–37.
-
Bardou M, Barkun AN, Martel M. Obesity and colorectal cancer. Gut [Net]. 2013;62:933 LP–947.
-
Schulfer A, Blaser MJ. Risks of antibiotic exposures early in life on the developing microbiome. PLoS Pathog [Internet]. 2015;11:e1004903–e1004903.
-
Axelsson M. Study on personality and adherence to antibody therapy: a population-based report. BMC Psychol. 2013;1:3–viii.
-
StataCorp. Stata Statistical Software: Release 15. College Station: StataCorp LLC; 2017.
Acknowledgements
Nosotros wish to give thanks PCCIUR, Academy of Aberdeen, especially Artur Wozniak, for extracting the data and performing case-control matching.
Funding
This work is supported past Cancer Research U.k. (reference C37316/A25535).
Author information
Affiliations
Contributions
Study concept and design: all authors. Statistical analysis and interpretation of data: RM and CC. Literature review: SP. Initial drafting of the manuscript: SP and RM. Data interpretation and editing of the manuscript: SP, RM, CC, PM, CM and LS. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
The report was canonical past the Research Applications and Data Management Team at the University of Aberdeen and Queen'south University Belfast, Schoolhouse of Medicine, Ideals Committee (reference number: 18.02v2). According to Caldicott principles patients registered at participating practices were made aware that their bearding data could be used in research and had the opportunity to withdraw should they wish. The report was performed in accord with the Declaration of Helsinki.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Boosted data
Publisher'south note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary data
Rights and permissions
Open Access This article is licensed nether a Creative Commons Attribution four.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(due south) and the source, provide a link to the Artistic Commons license, and bespeak if changes were made. The images or other third political party material in this article are included in the commodity'due south Creative Commons license, unless indicated otherwise in a credit line to the cloth. If material is not included in the article's Creative Eatables license and your intended use is not permitted by statutory regulation or exceeds the permitted use, yous will demand to obtain permission direct from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/past/4.0/.
Reprints and Permissions
Most this article
Cite this article
McDowell, R., Perrott, Southward., Murchie, P. et al. Oral antibody use and early-onset colorectal cancer: findings from a case-control report using a national clinical database. Br J Cancer (2021). https://doi.org/10.1038/s41416-021-01665-seven
-
Received:
-
Revised:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1038/s41416-021-01665-7
Source: https://www.nature.com/articles/s41416-021-01665-7?error=cookies_not_supported&code=b51ef064-e13f-431b-ba07-34d8c7cc3ed7
0 Response to "A Nurse Is Teaching a Client Who Has a Family History of Colorectal Cancer"
Post a Comment